Antitumor effect of a nutrient mixture on colon cancer cells
Colorectal cancer is the second most deadly cancer in the United States and there is an urgent need to develop effective preventive and therapeutic strategies against it. The aim of our study was to investigate the antitumor effects of a nutrient mixture containing ascorbic acid, lysine, proline and green tea extract that has been previously demonstrated a strong anticancer activity in several cancer cell types. We studied the effect of NM on human colon cancer cell line HCA-7 in vitro by measuring cancer cell proliferation, invasion, and expression of pro-inflammatory COX-2 and COX-1 proteins. Human colon cancer cell line HCA-7 was cultured in RPMI medium supplemented with fetal bovine serum and antibiotics. After 24 h, the cells were treated with NM at 0, 50, 100, 500 and 1000 μg/ml, in triplicate at each dose. Cell proliferation was evaluated by MTT assay, invasion through Matrigel™, MMP secretion was evaluated by zymography, and COX-2 and COX-1 expression by Western blot. The results showed that NM significantly inhibited the growth of HCA-7 cells, by 61% at 500 μg/ml and 82% at 1000 μg/ml. HCA-7 cell invasion through Matrigel™ was significantly reduced by 90% at NM dose 50 μg/ml, and the invasion was virtually completely blocked at 100 μg/ml NM. NM also inhibited COX-2 expression in a dose-dependent fashion, but had no effect on COX-1 expression. Conclusions: our results suggest that NM has potent inhibitory effects on HCA-7 cell growth and expression of markers of inflammation of colon cancer, and can be used as a novel cancer treatment strategy.
Introduction
Colorectal cancer is the second most deadly cancer in the United States. The American Cancer Society estimates that approximately 51,020 Americans will die from the disease in 2019.1 Colon cancer affects both men and women over age 50 with approximately the same frequency. In addition to the advanced age, there are other risk factors associated with colorectal cancer such as family history, inflammatory bowel disease, diets rich in saturated fat, lack of exercise and obesity. However, more than 75% of colorectal cancer cases occur in individuals without any specific predisposing factor. While colorectal cancer is treatable upon early detection, once the cancer metastasizes to the lymph, liver, or other organs, the 5-year survival is less than 10%; most of these fatalities are associated with metastasis.2 Improved screening methods have been linked to the recent decline in incidence and mortality of colorectal cancer.
Several plant-based phytochemicals are increasingly being used in cancer treatments due to their antitumor actions, including induction of apoptosis. Our earlier studies demonstrated significant anticancer efficacy of the nutrient mixture (NM)—containing ascorbic acid, lysine, proline and green tea extract—by targeting multiple pathways in cancer progression and metastasis.3 In the current study, we investigated the antitumor potential of the nutrient mixture (NM) on human colon cancer cell line HCA-7 in vitro by measuring cell proliferation, Matrigel™ invasion, and the effect of NM on markers of inflammation: cyclooxygenase COX-1 and COX-2 protein expression.
Materials and Methods
Cell culture and composition of NM
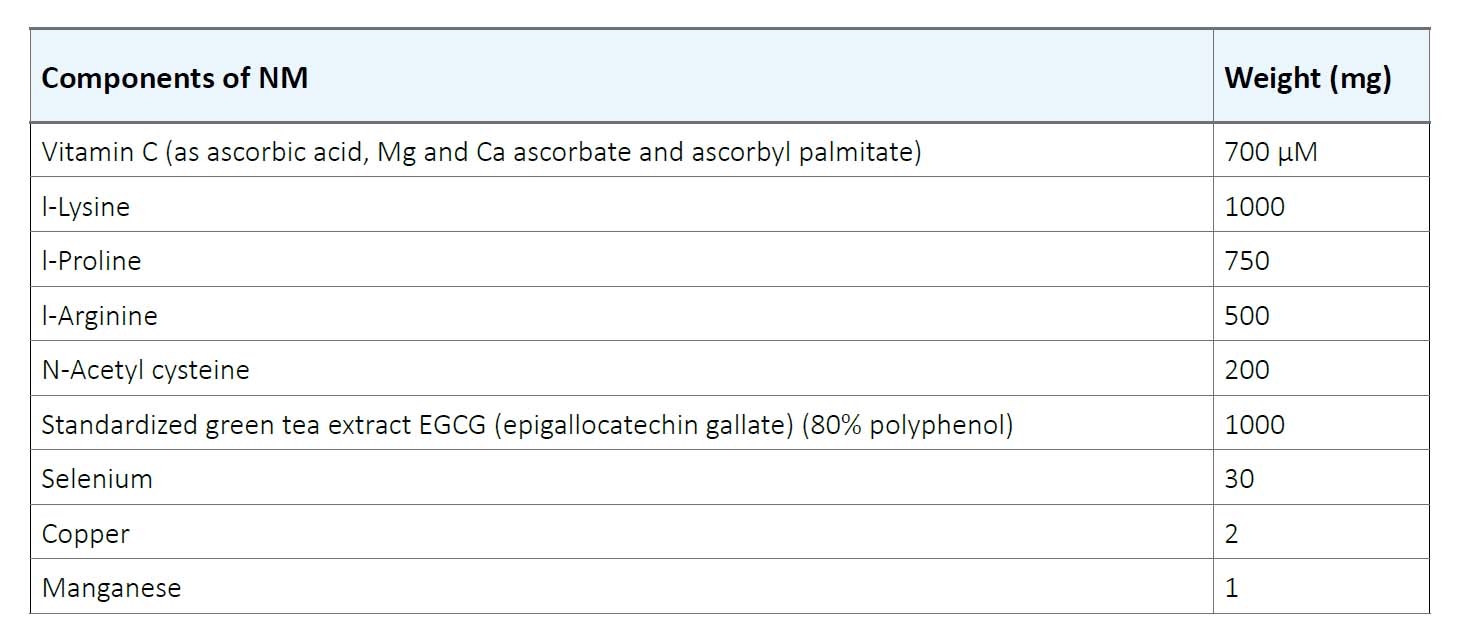
Human colon cancer cell line HCA-7 was purchased from ATCC (American Type Culture Collection) (Rockville, MD). The cells were cultured in RPMI medium supplemented with 10% FBS and antibiotics (100 U/mL penicillin and 100 μg/ml streptomycin) at 37°C in 5% carbon dioxide, 95% oxygen in a humidified cell incubator. The media was changed every alternate day. Cells were plated in 24-well tissue culture plates at a density of 3 x 104 cells until confluent. Then the cells were treated with NM at concentrations of 0, 50, 100, 500, and 1000 μg/ml in triplicate. The composition of NM is indicated in Table 1.
Table1. Nutrient Composition of Nutrient Mixture (NM)
MTT cytotoxic assay
Cell viability was determined using MTT assay. The assay is a colorimetric assay based on the ability of the viable cells to reduce soluble yellow tetrazolium salts to blue formazine crystals. After incubation of the cells with NM for 24 hours, 50μL of MTT reagent in media (0.5mg/ ml) was added to each well, and further incubated for 2 hours at 37°C. The solution was carefully aspirated and the blue formazine precipitate was dissolved in 1 ml DMSO and the absorbance (OD) was measured on a microplate reader at a wavelength of 570 nm in a BioSpec 1601 Shimadzu spectrometer. The percentage of inhibition was calculated.
Gelatinase zymography
Gelatinase zymography was performed in 10% Novex Pre-Cast SDS Polyacrylamide Gel (Invitrogen Corporation) in the presence of 0.1% gelatin under non-reducing conditions. Culture media (20 μl) were mixed with sample buffer and loaded for SDS-PAGE with tris-glycine SDS buffer, as suggested by the manufacturer (Novex). Samples were not boiled before electrophoresis. Following electrophoresis, the gels were washed twice in 2.5% Triton X-100 for 30 minutes at room temperature to remove the SDS. The gels were then incubated at 37°C overnight in a substrate buffer containing 50mM Tris-HCl and 10mM CaCl2 at pH 8.0, stained with 0.5% Coomassie Blue R250 in 50% methanol and 10% glacial acetic acid for 30 minutes and then destained. Upon renaturation of the enzyme, the gelatinases digested the gelatin in the gel, producing clear bands against an intensely stained background. Protein standards were run concurrently, and approximate molecular weights were determined by plotting the relative mobilities of known proteins.
H&E Staining
The HCA-7 colon cancer cells were cultured in 24-well plates and were treated with NM in test concentrations at 0, 50, 100, 500 and 1000 μg/ml, respectively. After 24-hour incubation, the cells were washed with phosphatebuffered saline (PBS), fixed with methanol, and then stained with hematoxylin and eosin (H&E), and images were captured by microscope.
Matrigel™ invasion
Invasion studies were conducted using Matrigel™ (Becton Dickinson) inserts in 24-well plates. HCA-7 cells were suspended in the medium and supplemented with NM, and seeded on the insert in the well. Thus, both the medium on the insert and in the well contained the same supplements. The plates with the inserts were then incubated in a culture incubator equilibrated with 95% air and 5% CO2 for 24 hours. After incubation, the media from the wells was withdrawn. The cells on the upper surface of the inserts were gently scrubbed away with cotton swabs. The cells that had penetrated the Matrigel™ membrane and migrated onto the lower surface of the Matrigel™ were stained with hematoxylin and eosin and visually counted under the microscope.
COX activity
COX-1 and COX-2 proteins were determined by Western blot analysis using polyclonal antibodies against COX-1 and -2 (Santa Cruz Biotechnology, Inc, Santa Cruz, CA). Briefly, HCA-7 cells were grown, challenged with NM dissolved in media at 0, 50, 100, 500, and 1000 μg/ml and incubated for 24 hours. The cells were then lysed for 30 min in ice-cold radioimmunoprecipitation (RIPA) buffer (50 mM Tris-HCl, pH 7.5, 10% Triton X-100, 1.0% Na-deoxycholate, 150 mM NaCl, 2 mM EDTA) (Teknova) with 1mM activated orthovanadate, and 1x Complete Mini Protease Inhibitor Cocktail tablet (Roche). Clarified cell lysates were denatured and equal proteins loaded and fractionated by SDS-polyacrylamide gel electrophoresis. After electrophoresis, the proteins were transferred to nitrocellulose membrane. The membranes were then incubated with the respective antibodies and developed by the enhanced chemiluminescence system (ECL, Amersham Pharmacia Biotech).
Statistical analysis
The results were expressed as means ± SD, as indicated in the results. Data was analyzed by independent sample t-test.
Discussion
Cell proliferation
As shown in figure 1, the NM displayed significant inhibitory effect on growth of HCA-7 colon cancer cells in vitro in a dose-dependent manner, starting from NM concentrations of 100 μg/ml (25% inhibition) and reaching 82% inhibition of cell growth at 1000 μg/ml.
MMP Expression by Zymography
Gelatinase Zymography study did not show any bands indicating the secretion of MMP-2 or MMP-9 (Figure not shown).
Cell Morphology by H&E staining and apoptosis
Morphology of the cells cultured for 24 hours in the presence of NM was evaluated by H&E staining and microscopy. H&E staining of HCA-7 cells exposed to the experimental concentrations of NM showed dose-dependent morphology changes with increasing concentrations of NM doses, with mild changes seen at 50 μg/ml, to significant apoptotic changes observed at 1000 μg/ml. (Figure 2A-2E) These include shrinking of the cytoplasm, darkly stained nuclei and intensely acidophilic cytoplasm— all of which suggest that cells are undergoing apoptosis.
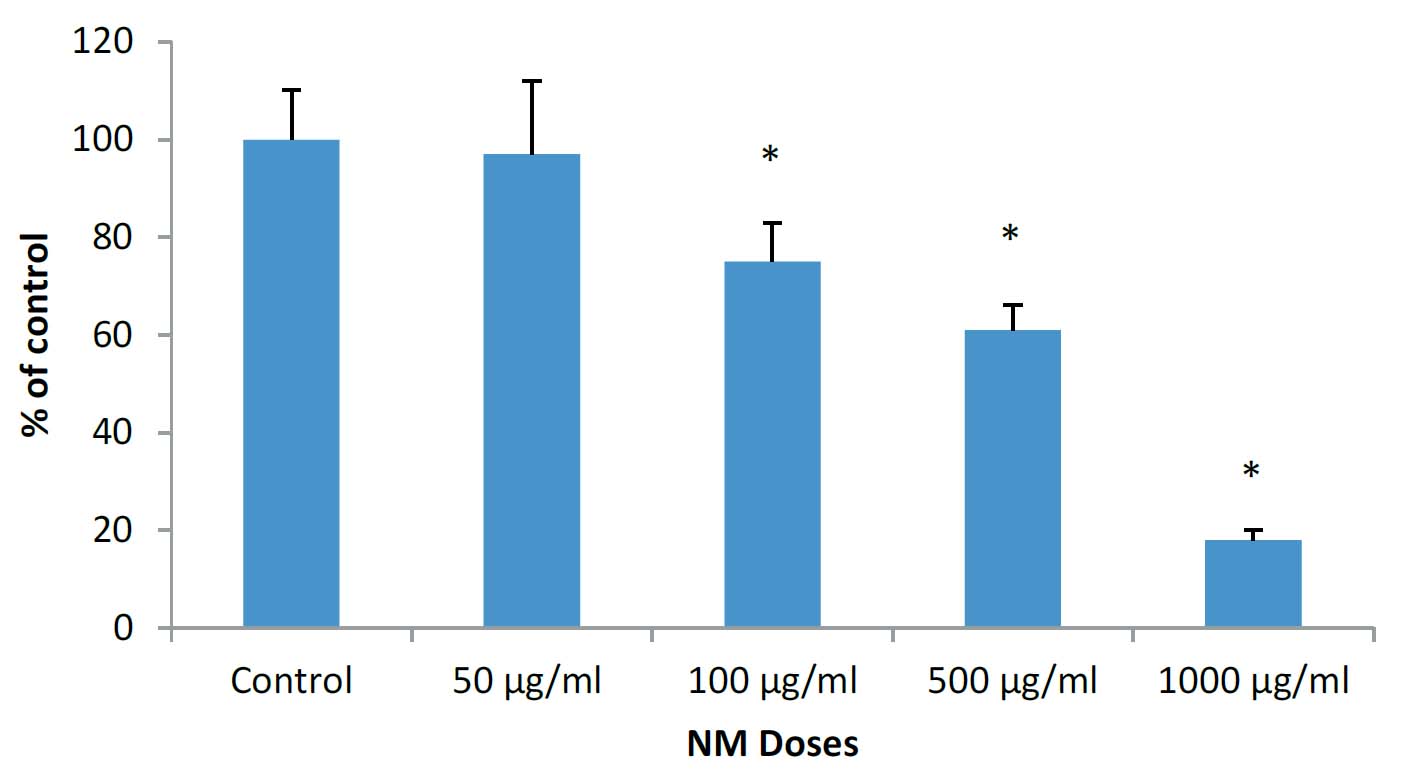
Figure 1: Effect of NM on cell proliferation of HCA-7 colon cancer cells: MTT assay (*significance=0.01)
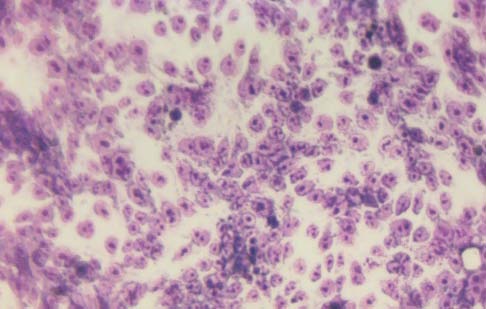
2A-Control
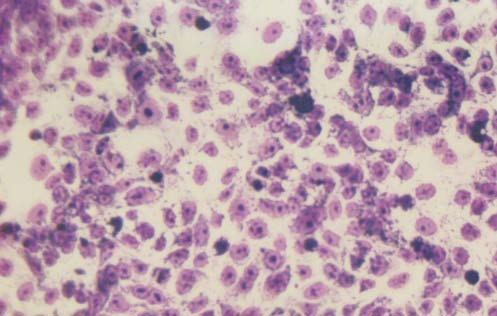
2B- NM 50 μg/ml
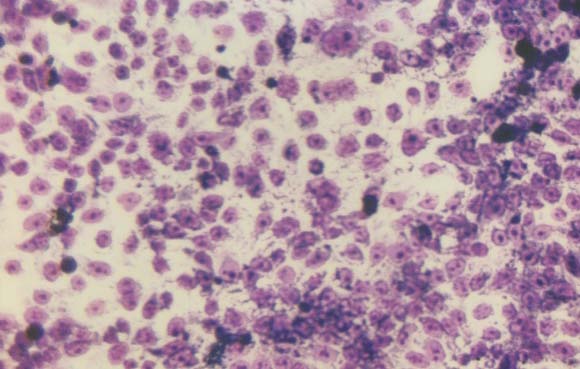
2C- NM 100 μg/ml
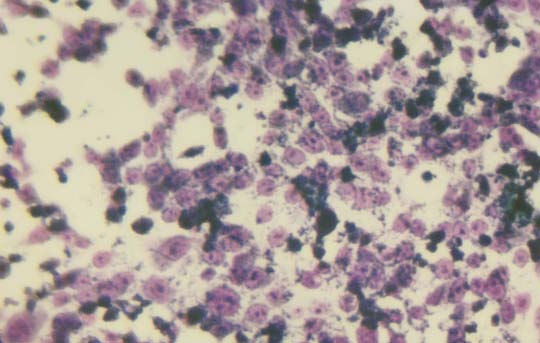
2D- 500 μg/ml
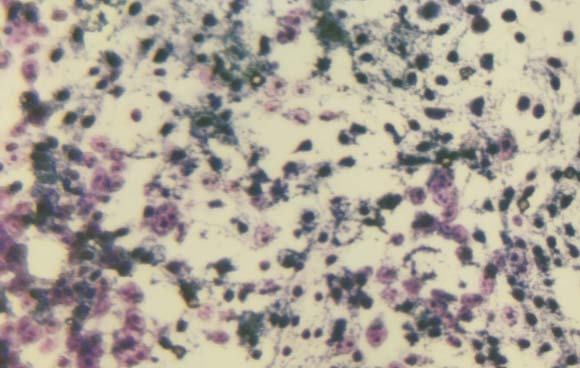
2E 1000 μg/ml
Figure 2: Morphology of HCA-7 cells exposed to different NM concentrations
Matrigel™ invasion
NM significantly inhibited the Matrigel™ invasion of HCA-7 colon cancer cells. Ninety percent inhibition was seen at 50 μg/ml, and virtually 100% inhibition of cell migration was seen at 100μg/ml, as shown in figure 3A. Figures 3B-3D present the photomicrographs of HCA-7 Matrigel™ invasion under microscope.
COX-1 and COX-2 expression
NM showed significant inhibitory effect on COX-2 expression by HCA-7 colon cancer cells. The COX-2 expression at 50 and 100 μg/ml was comparable to that of control, however it decreased gradually with higher NM doses up to 1000 μg/ml. (Figure 4A). The results on Figure 4B show that NM at tested concentrations did not affect the expression of COX-1.
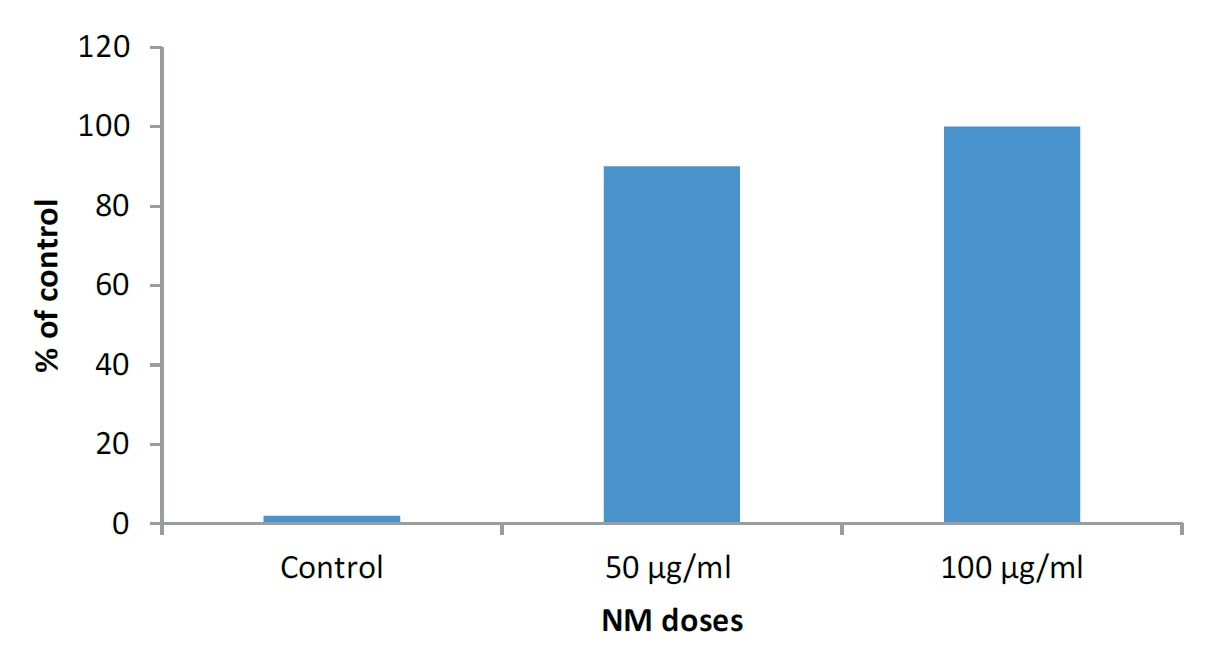
Figure 3A: Effect of NM on invasion HAC-7 colon cancer cells through Matrigel™
3B- Control
3C-NM 50 μg/ml
3D- 100 μg/ml
Figure 3B-3D: Photomicrographs of HCA-7 Matrigel™ Invasion
4A- Activity of COX-2
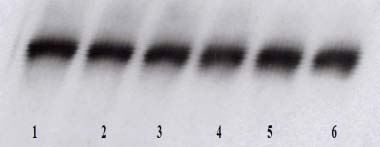
4B- Activity of COX-1
Figure 4: Effect of NM on HCA-7 cells COX-2 and COX-1 enzymatic expression (Legend: 1 – control; 2-6 – NM 50, 100, 250, 500, 1000 μg/ml)
Results
In the present study we investigated the effects of NM on human colon cancer cell line HCA-7. The results suggest that NM is effective in inhibiting the Matrigel™ invasion, cell proliferation and expression of inflammatory cyclooxygenase-2 (COX) enzymes in a dose-dependent fashion. In addition, NM also caused morphological changes, indicating cell apoptosis. HCA-7 colon cancer cells did not express MMP-2 or -9. These findings correlate with our previous studies which showed that zymography assay does not indicate the secretion of these MMP enzymes by HCA-7 cells.4
Invasion of cell basement membrane and extra cellular matrix (ECM) are the key steps in tumor progression and metastasis. The ECM invasion by cancer cells can be modulated by increased stability and strength of the connective tissue, secondary to the activity of the nutrients provided in NM. It is well known that ascorbic acid—a component of NM—is essential for the synthesis and hydroxylation of collagen fibers. Ascorbic acid (vitamin C) is not produced in the human body and low levels have been reported in cancer patients.5-7 The inhibitory effects of the individual nutrients composing NM in various aspects of cancer have been shown in both clinical and experimental studies. As such, ascorbic acid has been shown to have cytotoxic and antimetastatic effects on various malignant cell lines.8-10 Green tea extract is a potent anticancer agent and its inhibitory effects have been demonstrated in some human cancers. However, individual nutrients are not as effective as synergetic combinations, as we have shown in previous studies which demonstrated that anticancer effect of a combination of ascorbic acid, proline, lysine, and EGCG was greater than that of its individual components in several cancer cell lines. We have also demonstrated that the NM conferred superior anticancer effects compared to equimolar concentration of EGCG found in NM.11
Cyclooxygenase-2 (COX-2) is an enzyme overexpressed at sites of inflammation and in several epithelial cancers. COX-2-derived prostaglandins are involved in a variety of pathologic processes, including colorectal cancer. The study by Eberhart et al. linked increased expression of COX-2 to carcinogenesis.12 In addition to inflammation, a critical process in facilitating cancer cells growth and progression is angiogenesis. It has been shown that COX-2 is an important mediator of inflammation, angiogenesis and tumor growth. Modulation of COX-2 enzymes has been a promising field investigated by many research groups for prevention and treatment for colorectal cancer.
Early-stage colon cancer is generally treated with surgery, however—depending on the extent of excision of the tumor and surrounding tissues—the post-operative effects could be diarrhea, constipation, bleeding, infection, and sometimes a patient will require a permanent colostomy. Treatment of advanced colon cancer involves both chemotherapy and radiation therapy. Both of these approaches are associated with severe side effects. These treatment methods have not only been ineffective in providing a cure, but involve an indiscriminate attack on all cells, causing cellular damage and destruction of the body’s connective tissue, facilitating cancer metastasis. Clearly, there is a need for safe and effective therapeutic approaches that can be used to control the process of cancer metastasis as well as to prevent colon cancer.1, 2 Many studies confirm a direct correlation between COX-2 and colorectal cancer, indicating that inhibition of COX-2 is an effective pathway in preventing colon cancer.13, 14 However, some COX-2 inhibitors such as NSAIDs, have their own serious side effects and increased likelihood of bleeding tendencies.
In our study we have shown that NM can inhibit COX-2 expression in a dose-dependent fashion, along with inhibition of cancer cell growth and invasion. Our previous studies in various cancer cell lines have demonstrated that NM also has a potent anti-angiogenic activity.15 Earlier we demonstrated that NM applied even at high concentrations is non-toxic.16 Although efforts continue to explore novel uses of NSAIDs and other COX-2 inhibitors for colorectal cancer, our study indicates a wide potential of NM as a safe and effective option in colon cancer.
References
- American Cancer Society (www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html) Accessed Mar 2019
- National Cancer Institute. What you need to know about cancer of colon,(www.cancer.net/survivorship) Accessed Mar 2019
- International Society for Infectious Diseases. ProMEDmail Website. www.promedmail.org. Accessed April 29, 2004.
- Netke SP, Roomi MW, Ivanov V, Niedzwiecki A, Rath M. A specific combination of ascorbic acid, lysine, proline and epigallocatechin gallate inhibits proliferation
of extracellular matrix invasion of various human cancer cell lines. Res comm in Pharm Toxico: Emerging Drugs. 2003:2,37-50. - Roomi MW, Monterrey JC, Kalinovsky T, Rath M, Niedzwiecki A. Patterns of MMP-2 and MMP-9 expression in human cancer cell lines. Oncology Reports. 2009;21:1323-1333.
- Koh WS, Lee SJ, Lee H, Park C, Park MH, Kim WS, et al. Differential effects and transport kinetics of ascorbate derivatives in leukemic cell lines. Anticancer Res. 1998;8:2487-2493.
- Nunez C, Ortiz de Apodaca Y, Ruiz A. Ascorbic acid in the plasma and blood cells of women with breast cancer. The effect of consumption of food with an elevated
content of this vitamin. Nutr Hosp. 1995;10:68-372. - Kurbacher CM, Wagner U, Kolster B, Andreotti PE, Krebs D, Bruckner HW. Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin
and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett. 1996;103(2):183-189. - Roomi MW, House D, Eckert-Maksic M, Maksic ZB, Tsao CS. Growth suppression of malignant leukemia cell line in vitro by ascorbic acid (vitamin C) and its
derivatives. Cancer Lett. 1998;122:93-99. - Naidu KA, Karl RC, Coppola D. Antiproliferative and proapoptotic effect of ascorbyl stearate in human pancreatic cancer cells: association with decreased
expression of insulin-like growth factor 1 receptor. Dig Dis Sci. 2003;48(1):230-7. - Anthony HM, Schorah CJ. Severe hypovitaminosis C in lung cancer patients: The utilization of vitamin C in surgical repair and lymphocyte-related host resistance.
Br J Cancer 1982;46:354-367. - Roomi MW, Monterrey JC, Kalinovsky T, et al. Comparative effects of EGCG, green tea and a nutrient mixture on the patterns of MMP-2 and MMP-9 expression in cancer cell lines. Oncol Rep 2010;24:747–57.
- Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology. 1994;107(4):1183-8.
- Sheng H, Shao J, Kirkland SC. Inhibition of human colon cancer cell growth by selective inhibition of cyclooxygenase-2. J Clin Invest. 1997;99:2254-2259.
- Williams CS, Sheng H, Brockman JA. A cyclooxygenase-2 inhibitor (SC-58125) blocks growth of established human colon cancer xenografts. Neoplasia. 2001;3:428-436.
- Roomi MW, Roomi N, Ivanov V, et al. Inhibitory effect of a mixture containing ascorbic acid, lysine, proline, and green tea extract on critical parameters in angiogenesis. Oncol Rep. 2005;14:807-15.
- Roomi MW, Ivanov V, Netke SP, Niedzwiecki A, Rath M. Serum markers of the liver, heart, and kidney and lipid profile and histopathology in ODS rats treated with nutrient synergy. J. Am. Coll. Nutr. 2003;22:477.
Table 1. Nutrient Composition of Nutrient Mixture (NM)
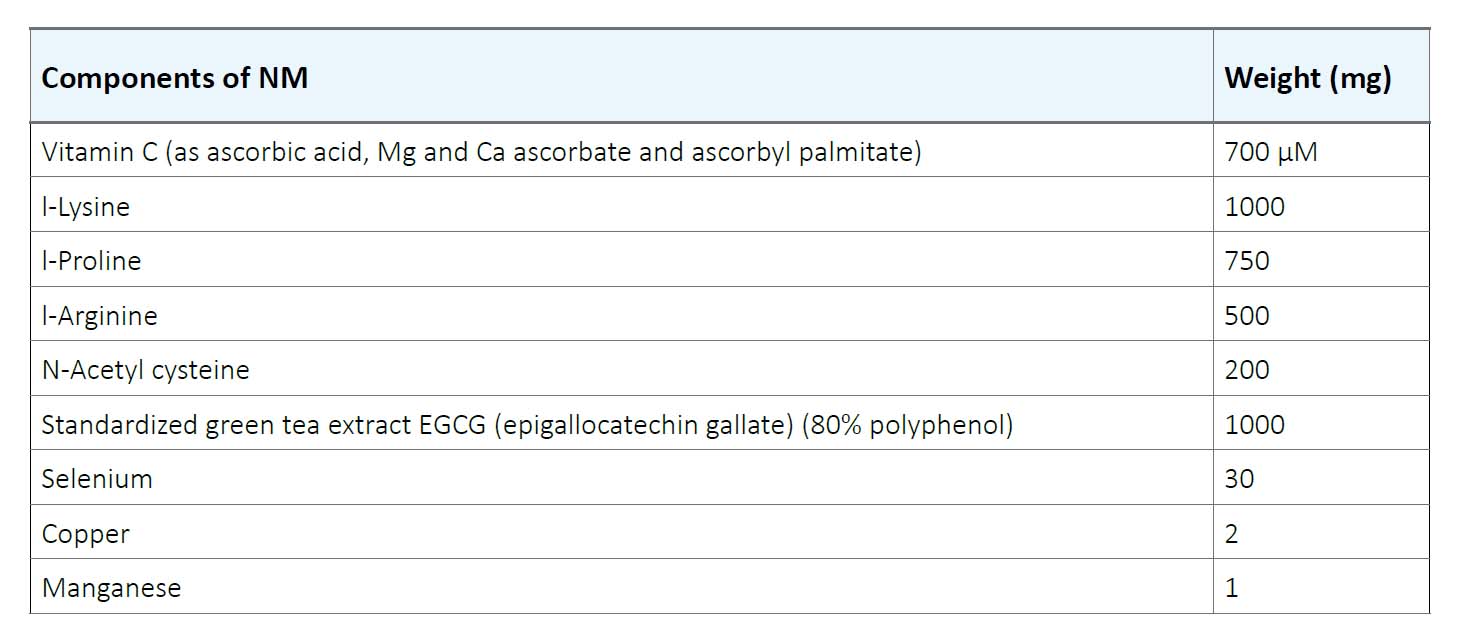

Figure 1: Effect of NM on cell proliferation of HCA-7 colon cancer cells: MTT assay (*significance=0.01)

2A-Control

2B- NM 50 μg/ml

2C- NM 100 μg/ml

2D- 500 μg/ml

2E 1000 μg/ml
Figure 2: Morphology of HCA-7 cells exposed to different NM concentrations

Figure 3A: Effect of NM on invasion HAC-7 colon cancer cells through Matrigel™

3B- Control

3C-NM 50 μg/ml

3D- 100 μg/ml
Figure 3B-3D: Photomicrographs of HCA-7 Matrigel™ Invasion

4A- Activity of COX-2

4B- Activity of COX-1
Figure 4: Effect of NM on HCA-7 cells COX-2 and COX-1 enzymatic expression (Legend: 1 – control; 2-6 – NM 50, 100, 250, 500, 1000 μg/ml)
References
- American Cancer Society (www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html) Accessed Mar 2019
- National Cancer Institute. What you need to know about cancer of colon,(www.cancer.net/survivorship) Accessed Mar 2019
- International Society for Infectious Diseases. ProMEDmail Website. www.promedmail.org. Accessed April 29, 2004.
- Netke SP, Roomi MW, Ivanov V, Niedzwiecki A, Rath M. A specific combination of ascorbic acid, lysine, proline and epigallocatechin gallate inhibits proliferation
of extracellular matrix invasion of various human cancer cell lines. Res comm in Pharm Toxico: Emerging Drugs. 2003:2,37-50. - Roomi MW, Monterrey JC, Kalinovsky T, Rath M, Niedzwiecki A. Patterns of MMP-2 and MMP-9 expression in human cancer cell lines. Oncology Reports. 2009;21:1323-1333.
- Koh WS, Lee SJ, Lee H, Park C, Park MH, Kim WS, et al. Differential effects and transport kinetics of ascorbate derivatives in leukemic cell lines. Anticancer Res. 1998;8:2487-2493.
- Nunez C, Ortiz de Apodaca Y, Ruiz A. Ascorbic acid in the plasma and blood cells of women with breast cancer. The effect of consumption of food with an elevated
content of this vitamin. Nutr Hosp. 1995;10:68-372. - Kurbacher CM, Wagner U, Kolster B, Andreotti PE, Krebs D, Bruckner HW. Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin
and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett. 1996;103(2):183-189. - Roomi MW, House D, Eckert-Maksic M, Maksic ZB, Tsao CS. Growth suppression of malignant leukemia cell line in vitro by ascorbic acid (vitamin C) and its
derivatives. Cancer Lett. 1998;122:93-99. - Naidu KA, Karl RC, Coppola D. Antiproliferative and proapoptotic effect of ascorbyl stearate in human pancreatic cancer cells: association with decreased
expression of insulin-like growth factor 1 receptor. Dig Dis Sci. 2003;48(1):230-7. - Anthony HM, Schorah CJ. Severe hypovitaminosis C in lung cancer patients: The utilization of vitamin C in surgical repair and lymphocyte-related host resistance.
Br J Cancer 1982;46:354-367. - Roomi MW, Monterrey JC, Kalinovsky T, et al. Comparative effects of EGCG, green tea and a nutrient mixture on the patterns of MMP-2 and MMP-9 expression in cancer cell lines. Oncol Rep 2010;24:747–57.
- Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology. 1994;107(4):1183-8.
- Sheng H, Shao J, Kirkland SC. Inhibition of human colon cancer cell growth by selective inhibition of cyclooxygenase-2. J Clin Invest. 1997;99:2254-2259.
- Williams CS, Sheng H, Brockman JA. A cyclooxygenase-2 inhibitor (SC-58125) blocks growth of established human colon cancer xenografts. Neoplasia. 2001;3:428-436.
- Roomi MW, Roomi N, Ivanov V, et al. Inhibitory effect of a mixture containing ascorbic acid, lysine, proline, and green tea extract on critical parameters in angiogenesis. Oncol Rep. 2005;14:807-15.
- Roomi MW, Ivanov V, Netke SP, Niedzwiecki A, Rath M. Serum markers of the liver, heart, and kidney and lipid profile and histopathology in ODS rats treated with nutrient synergy. J. Am. Coll. Nutr. 2003;22:477.

